The hierarchy of risk controls in dentistry
How to implement clinical risk containment and control methods
In recent years, the world of dentistry has undergone major changes: the rapid technological evolution of materials and equipment, the digitisation of processes, a new awareness of the patient, more attentive to results and costs, and an increasing focus on prevention and aesthetics. Under these conditions and in a post-pandemic context, the importance of paying attention to the issue of infection risk is immediately apparent. It has become necessary to implement actions to monitor, intercept and control the various clinical risk situations, in order to prevent adverse events, and to recognise and remove any risky procedures where such events have already occurred. Managing risks means analysing the sources of these adverse events, rating them, verifying whether the associated risk is acceptable or unsustainable, and in the latter case, identifying actions to eliminate or reduce it. In this context, it may be useful to keep in mind the hierarchy of risk controls, a system used to minimise or eliminate risk exposure.
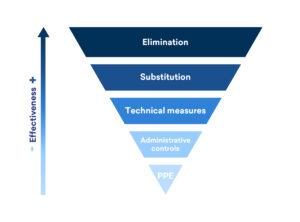
The hierarchy of infection control begins with the two most effective methods, namely risk elimination and substitution, which replaces potential risk factors with practices that are less likely to cause infection. In third place are technical controls, i.e. those mechanical instruments that can isolate the hazard from people. The next step in the hierarchy is the implementation of administrative controls, designed to protect the individual when the risk cannot be removed. Personal Protective Equipment (PPE) is the last level of defence in the hierarchy. For maximum efficacy, it requires the previous steps to be in place.A 2017 study1 found that in dental clinics, due to the high production of bioaerosols, workers are exposed to a high risk of contracting infectious diseases, and that this increases the likelihood of cross-infection. To maintain the safety of dentists, their families and patients, here are some simple ways to put the hierarchy of risk control into practice:
- Elimination
Elimination consists of removing the hazard. A prime example is when a patient calls the dental practice to say they are unwell, and their visit is postponed until they feel better.
2. Substitution
However, elimination is not always a viable option. A viable alternative may be to replace it with a safer solution. Since every patient must be considered potentially infected, some replacements may reduce the risk of exposure, while helping the patient. A good option is the use of minimally invasive protocols to reduce the production of splashes, sprays and aerosols. Among the instruments that enable the implementation of minimally invasive protocols are surgical aspirator tips, which can reduce aerosol production by up to 90%, and dental dams, which create an effective barrier between the area to be treated and the rest of the mouth, thus limiting contamination by bacteria present in the oral cavity.
3. Technical Measures
Technical measures consist of the use of barrier systems to isolate the hazard from people. In the dental world, many devices are used that may fall into the third level of the control hierarchy, including sharps containers, devices that eliminate the possibility of needle punctures, and ultrasonic tanks to reduce the bacterial load on instruments compared to manual rubbing of sharp instruments. All these devices have been developed to reduce the possibility of exposure to pathogens.
A further example of a technical measure is the use of ventilation systems in the dental clinic, which purify the outside air by filtering out microbes and release the used air through a separate exhaust duct. Most essential dental procedures produce contaminated aerosols, splashes or sprays, so it is crucial to control and contain these fluids before they reach patients and start travelling through the air.
4. Administrative controls
Administrative controls, also known as work practice controls, are employer-dictated measures, i.e. policies and procedures that change the way the dental team works in order to reduce or avoid risk exposure. For example, if you have an aspirator tip and it is recommended for use during procedures with splash or aerosol production, the work practice control consists of checking that it is actually used. Having a container for sharps but then throwing a needle in the rubbish, on the other hand, is an example of implementing technical measures without adequate controls on working practices.
Standard operating procedures are useful to ensure that work practice controls are implemented, because these procedures can help employees quickly visualise and understand processes for tasks, even in complicated procedures, thus eliminating repetitive questions. Establishing systems and best practices can also foster accountability among staff, which helps establish behaviours that will ultimately keep everyone safe.An example of great importance in managing risk by defining and verifying shared procedures and protocols is the reconditioning of instruments. Because this process consists of several steps (cleaning, disinfection, sterilization, packaging and storage), it should always be introduced by a training phase on the protocol and accompanied by instructions on the good practices to be followed and the relevant checks to be conducted.
5. Personal Protective Equipment (PPE)
The hierarchy of controls ranks the categories of risk controls from most effective to least effective, and PPE is the last recommended line of defence. Although PPE is a fundamental protection mechanism, we cannot expect it to be our only defence, nor an infallible mechanism. Therefore, all the other risk control steps listed above must be implemented in addition to the use of PPE. There is currently much talk of PPE, a general term for any kind of Personal Protective Equipment. From a regulatory point of view, however, there are two different types of devices: Medical Devices and Personal Protective Equipment. The latter is defined by EU Regulation no. 425/2016, and its function is to protect the wearer from health or safety risks. In a dental practice, this equipment includes: cut-proof gloves, a protective face mask, a waterproof gown and protective glasses.Medical Devices include dental and orthodontic prosthetics, implants, disinfectants, dental chairs and autoclaves. All Medical Devices must be certified and comply with the new European MDR Regulation, unlike Medical Devices, is therefore designed to protect the wearer from hazards. After each patient, face mask and gloves must be replaced. Furthermore, if a protective device – be it a Medical Device or PPE – is visibly dirty, wet or torn, it must be replaced.
When patients contract diseases in dental practices, these cases are considered healthcare-associated infections and have consequences for those responsible, who sometimes underestimate infection control and the possible financial penalties they may face.
The solution is to remain up-to-date with the latest regulations and recommendations and to diligently implement all levels of the risk control hierarchy.
Awareness of the risks associated with their professional activities gives dentists greater confidence in the management of clinical practices. The risk analysis highlights the operations that are most likely to be repeated in order to obtain the result, suggesting possible corrections and highlighting the need to eliminate behaviour that may be damaging (in an economic, aesthetic or physical sense) to the patient or to the dental practice.




